Stem cell therapy - future therapeutic in regeneration medicine
Stem cell therapy has been one of the most significant cell therapeutic strategies for researchers to utilize as a treatment for various range of diseases. Stem cell therapy has great potential for future therapeutic uses in tissue regeneration and repair. They have the potential to build every tissue in the human body by differentiating it into specific cell types.
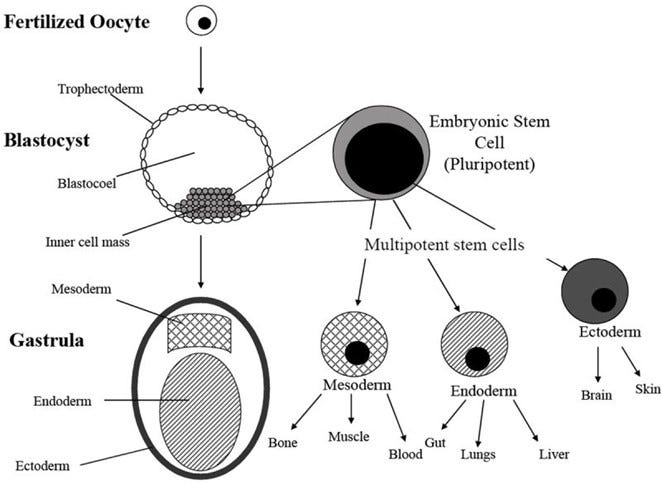
For a cell to be considered a “stem cell”, two characteristics are important: cells must have the ability of unlimited self-renewal to produce progeny exactly the same as the originating cell in a highly controlled manner, and that they must be able to give rise to a specialized cell type. Stem cells can be differentiated into two types: pluripotent and multipotent. Pluripotent stem cells have the ability to differentiate into all cell types in the body. Multipotent stem cells will go through a series of divisions to give rise to even more restricted specialized tissues and cells.
Image Source: Introduction to Stem Cell Therapy - PMC (nih.gov)
Stem cells can be further divided into three categories: embryonic, germinal, and somatic.
Embryonic stem cells originate from the inner cell mass of the blastocyte and are omnipotent. Germinal stem cells originate from the primary germinal layer of embryo and further differentiate into progenitor cells to produce specific organ cells. Somatic/adult stem cells are progenitor cells that exist in hematopoietic, neural, gastrointestinal, and mesenchymal tissues. Somatic/adult stem cells from bone marrow (hematopoietic and mesenchymal stem cells) are the most commonly used stem cells as a therapeutic treatment. Peripheral blood also provides a safe and easily accessible route for isolating adult stem cells that has shown to induce more T and NK (natural killer) cells compared to bone marrow adult stem cells.
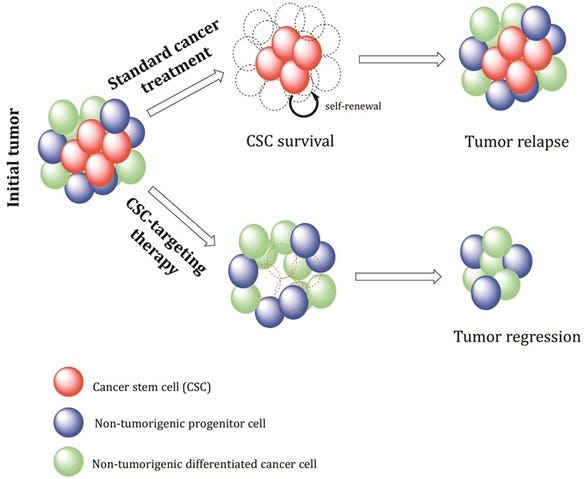
For over 30 years, stem cells have been used in the replenishment of blood and immune systems damaged by the cancer cells or during the treatment of cancer by chemotherapy or radiotherapy. Traditional therapies against cancer such as chemotherapy and radiotherapy, have limitations that lead to treatment failure and cancer recurrence. These limitations are related to systemic and local toxicity, while treatment failure and cancer recurrence is due to drug resistance and self-renewal properties of small population of cells called – “cancer stem cells.” Cancer stem cells can be defined as cells in the tumor growth with a tumor initiating potential. One major difference between normal stem cells and cancer stem cells is that there is no control on divisions of cells in cancer stem cells. The cancer stem cells have been shown to be not only capable of self-renewal but also generating a wide spectrum of progeny, like normal stem cells. In order to develop efficient treatments that can induce a long-lasting clinical response preventing tumor relapse, it is important to develop drugs that can specifically target and eliminate cancer stem cells. The combined therapy using conventional anticancer drugs with cancer stem cell targeting agents, may offer a promising strategy for management and eradication of various types of cancers.
Image Source: Pharmacology | CRS | Paul Scherrer Institute (PSI)
Clinical utilities
There are many clinical trials going on related to stem cell therapies. In a recent Phase I clinical trial, intrauterine injection of clinical graded human umbilical cord derived mesenchymal stem cells were used for poor healing after uterine injury. After Phase I trials, the therapy was considered safe for poor healing after uterine injury and will be further examined in the next set of clinical trials. Mesenchymal stem cell treatment has also been used in a study in patients with Perianal Fistulizing Crohn’s disease. An extended follow up study of adipose derived mesenchymal stem cells for induction of remission in perianal fistulizing Crohn’s disease or ADMIRE-CD Phase 3 clinical trial was carried out last year to evaluate the long-term safety and efficacy of Darvadstrocel (expanded allogenic adipose derived stem cell therapy). The study showed no safety concerns up to 104 weeks after the treatment and that the clinical remission after a single administration of Darvadstrocel may be sustained in the long term (up to 104 weeks).
Stem cell therapies are showing increasing promise in the treatment of cancer. They can function as novel delivery functions homing to and targeting both primary and metastatic tumor foci. Stem cells engineered to stably express various cytotoxic agents decrease tumor volumes and extend survival in preclinical animal models. Stem cells can also be applied in regenerative medicine, immunotherapy, cancer stem cell targeted therapy, and anti-cancer drug screening applications. Even though stem cells provide a feasible solution to treat human cancers, challenges such as treatment durability and tumorigenesis make it necessary to improve therapeutic performance and applicability. Conquering the limitations of stem cell-based therapies by doing some additional research is important for more widespread utilization of stem cell therapies.
References
Biehl, J. K., & Russell, B. (2009). Introduction to Stem Cell Therapy. Journal of Cardiovascular Nursing, 24(2), 98–103. https://doi.org/10.1097/jcn.0b013e318197a6a5
Denisa, Dragu, Laura, Necula, Coralia, Bleotu, Carmen, Diaconu, Mihaela, & Chivu-Economescu. (2015). Therapies targeting cancer stem cells: Current trends and future challenges. 世界干细胞杂志:英文版(电子版), 9, 1185–1201. http://www.cqvip.com/QK/71424X/201509/87748367504849534857484851.html
García-Olmo, D., Gilaberte, I., Binek, M., D´Hoore, A. J., Lindner, D., Selvaggi, F., Spinelli, A., & Panés, J. (2022). Follow-up Study to Evaluate the Long-term Safety and Efficacy of Darvadstrocel (Mesenchymal Stem Cell Treatment) in Patients with Perianal Fistulizing Crohn’s Disease: ADMIRE-CD Phase 3 Randomized Controlled Trial. Diseases of the Colon & Rectum, 65(5), 713–720. https://doi.org/10.1097/dcr.0000000000002325
Huang, J., Li, Q., Yuan, X., Liu, Q., Zhang, W., & Li, P. (2022). Intrauterine infusion of clinically graded human umbilical cord-derived mesenchymal stem cells for the treatment of poor healing after uterine injury: a phase I clinical trial. Springer Link, 13(1). https://doi.org/10.1186/s13287-022-02756-9
Sagar, J., Chaib, B., Sales, K. M., Winslet, M. C., & Seifalian, A. M. (2007). Role of stem cells in cancer therapy and cancer stem cells: a review. Cancer Cell International, 7, 9. https://doi.org/10.1186/1475-2867-7-9
Zhang, C., Huang, T., Wu, B., He, W., & Liu, D. (2017). Stem cells in cancer therapy: opportunities and challenges. Oncotarget, 8(43), 75756–75766. https://doi.org/10.18632/oncotarget.20798