Adoptive cell therapies (ACT) have emerged as a promising therapeutic option in cancer treatment, with Tumor-Infiltrating Lymphocyte (TIL). TIL therapy involves extracting immune cells from a patient's tumor tissue, expanding them ex vivo, and infuse them back into the patient to target and eliminate cancer cells. Recent research advancements in TIL therapy highlight its potential immuno-therapeutic strategy.
Improving TIL Selection and Expansion:
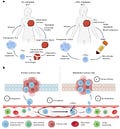
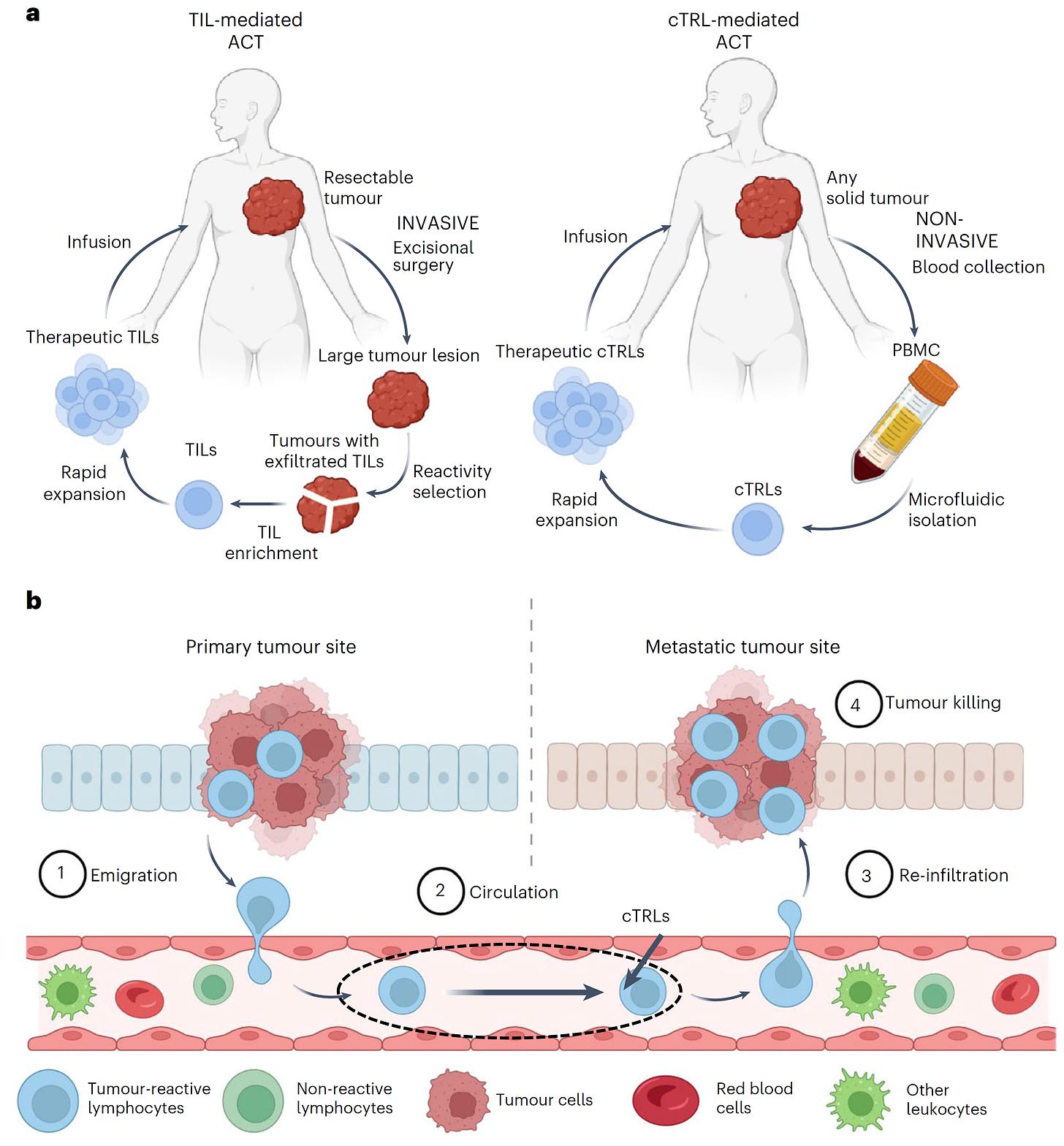
Efficient selection and expansion of TILs are crucial for successful clinical outcomes. A recent study has focused on circulating tumor-reactive lymphocytes (cTRLs) can be isolated from peripheral blood at high yield and purity via microfluidic immunomagnetic cell sorting (Wang, et al., Nature Biomedical Engineering, 2023). One notable advancement is the use of peripheral blood instead of obtaining large and fresh tumor tissues, which is often not feasible in patients with unresectable tumors or recurrent metastasis. cTRLs exhibited comparable killing potency to TILs in vitro. Both cTRLs and TILs isolated from solid tumors extended median survival by 40 -50 % in vivo mouse model. Although the TCR repertoire of the cTRLs was not as diverse as the TILs, the feasibility of cTRLs treatment was verified with metastasis model.
Figure 1: Non-invasive collection of tumor reactive cells in peripheral blood. a. Conventional invasive TIL therapy (left) compared with non-invasive circulating tumor reactive lymphocytes (cTRLs, right). b, the trafficking dynamics of cTRLs in primary tumor site (left) and metastatic tumor site (right). (Wang, et al., Nature Biomedical Engineering, 2023)
Overcoming Tumor Microenvironment Suppression:
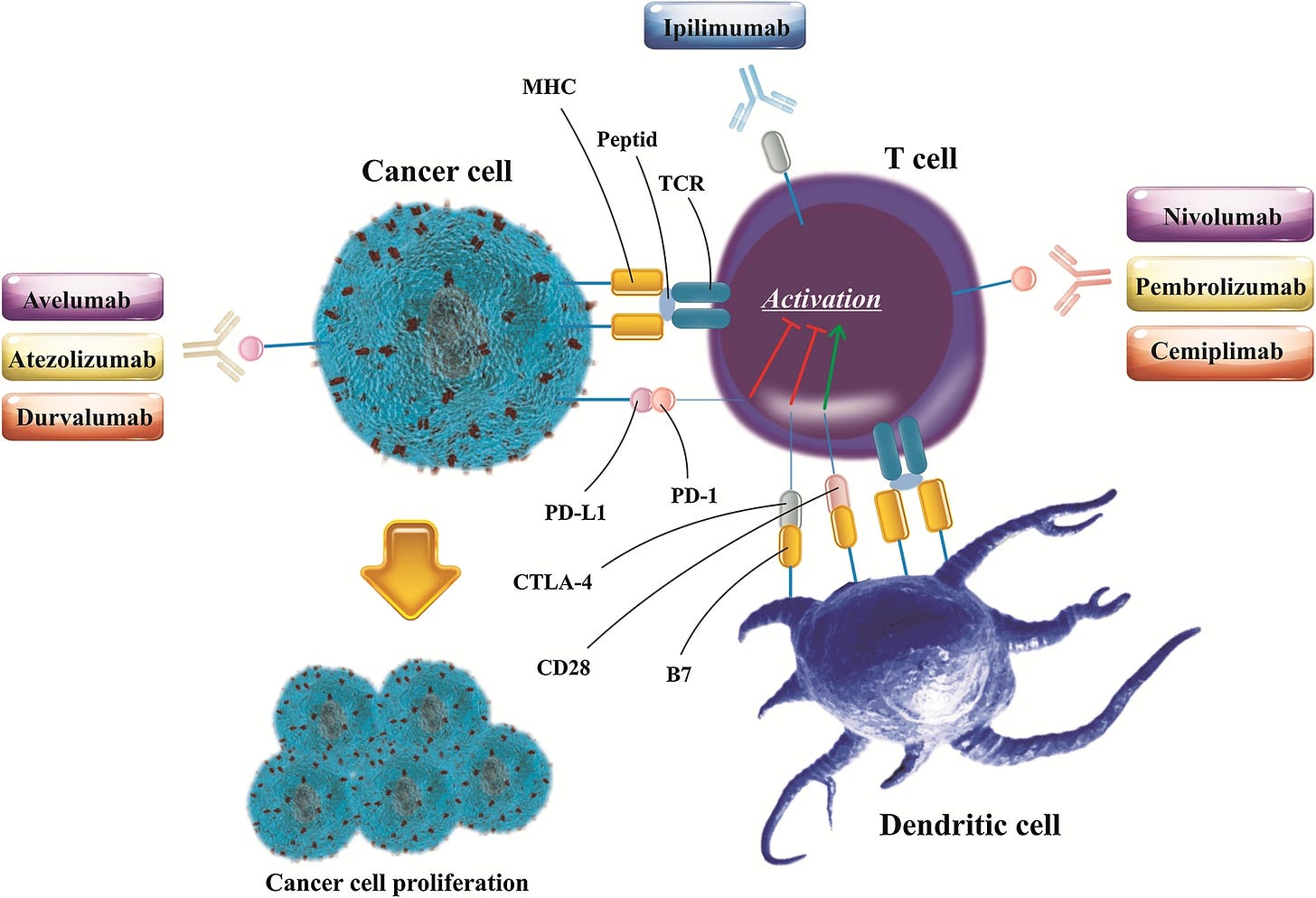
The tumor microenvironment (TME) presents a major obstacle to TIL therapy success, as it often creates an immunosuppressive milieu that hampers TIL effector function. Recent research has focused on developing strategies to reduce the immunosuppressive effects in TME. One of strategies is the combination of TIL therapy with immune checkpoint inhibitors, such as ipilimumab. It has shown promise in augmenting T cell activity and overcoming TME-mediated immune evasion. This combination approach using ipilimumab plus TILs has induced significant ORR (37.5%) and promoted PFS (7.3 months) in metastatic melanoma patients (Mullinax, et al., Front Oncol. 2018). Similarly, nivolumab plus TILs has indicated significantly higher ORR (33%), prolonged mPFS (5.4 months), and mOS (15.2 months) compared to the group without nivolumab which recorded 6.67%, 3.8 and 6.6 months in patients with metastatic osteosarcoma (Li, et al., J Bone Oncol., 2020).
Figure 2: The FDA approved immune checkpoint inhibitors. PD-1 inhibitor: nivolumab, pembrolizumab, cemiplimab, PD-L1 inhibitor: atezolizumab, avelumab, and durvalumab, and CTLA-4 inhibitor: ipilimumab (Vafaei, et al., Cancer Cell International, 2022).
Engineering TILs for Enhanced Functionality:
Genetic engineering techniques have opened new avenues for improving TIL functionality. Recent studies have explored gene editing approaches, such as Clustered regularly interspaced short palindromic repeats-Cas (CRISPR-Cas9), to enhance TILs' tumor-killing potential. By editing specific genes involved in T cell suppression and exhaustion, researchers have been able to create TILs with improved cytotoxicity and resistance to immunosuppression. These CRISPR modified TILs did not alter the ex vivo expansion efficiency, immunophenotype, nor the TCR clonal diversity of TIL. (Fix, et al., J Immunother Cancer, 2022). Overall, genetically modified TILs hold great promise for enhancing the therapeutic efficacy of TIL therapy.
Conclusion:
Recent research in TIL therapy has brought promising advancements that address key challenges in the field. By improving TIL selection, overcoming the suppressive TME, and leveraging genetical engineering, researchers are significantly enhancing the therapeutic efficacy and durability of TIL therapy. While TIL therapy still faces hurdles on the path to clinical implementation, the rapid progress in this field holds great promise for the future of cancer immunotherapy. Continued research efforts and collaborations between scientists, clinicians, and biotech companies are absolutely necessary to refine TIL therapy and accelerate its translation into clinical practice, ultimately benefiting cancer patients worldwide.
Fresh Wind Biotechnologies team dedicates our efforts in the development of TIL therapies in clinical utilities.
Fresh Wind Biotech (FWB) has well-orchestrated in-house operations in both the U.S. (Houston, Texas) and China (Tianjin) spanning cell therapy R&D, PD, GMP and commercialization. Our U.S. facility is located in Houston, Texas minutes from the world-renowned Texas Medical Center which allows us to collaborate with some of the leading clinical cancer institutions in the world. Our U.S. facility can accommodate multiple cell therapy products including tumor organoids, TIL process development and informatics. Our cell therapy advances at our U.S. facility positions FWB for Phase I clinical trials in U.S for TIL therapies and related therapeutic products.
Our teams are experienced, motivated and well trained. We have leading experts in TIL product manufacturing, flow cytometry, tumor organoids and informatics. We constantly seek collaborations with top tier universities, and clinical groups. Our teams are continuously recruiting talents who have a passion to work in a fast pace environment and push technologies into life-saving medical products.