Savvy researchers are increasingly using Organoids to speed up their pre-clinical studies over animal models
Non-human primates are increasingly replaced by organoids in pharma RED
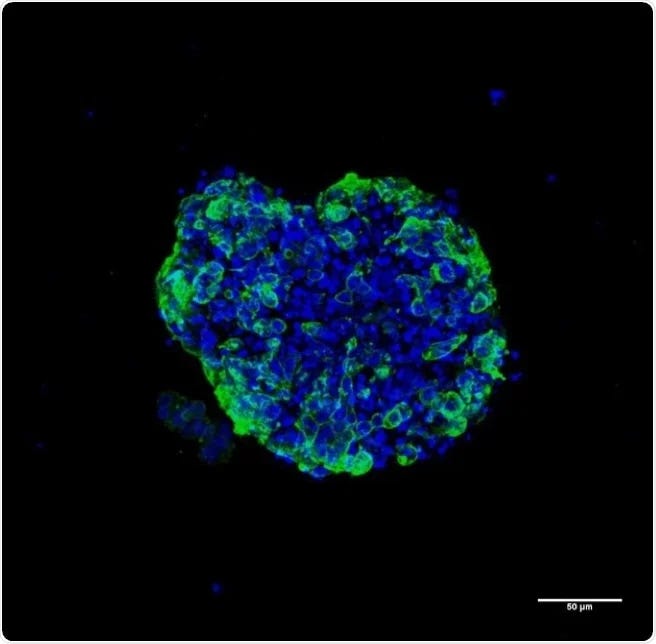
Savvy researchers are increasingly turning to tumor organoids to speed up their pre-clinical studies and bypassing the challenges and delays facing peers using traditional non-human primates (NHP) for pre-clinical studies.
(Pic source: https://www.nature.com/articles/d41586-021-01894-z)
Potential drug candidates are usually tested on animal models such as NHPs to support IND applications with the U.S. Food and Drug Administration (FDA) to commence clinical trials in humans. Over the last few years, a shortage of available NHPs due to increasing demand have led to delays and added expense in generating such data. This problem will continue to get worse based on recent news from Charles River Labs, one of the largest providers of NHPs for clinical research.
Non-human primate shortage will hurt pharma development
Charles River Labs recently reported that it has suspended future shipments of NHPs from Cambodia. CRA’s voluntary suspension is in reaction to a U.S. Department of Justice (USDOJ) subpoena about CRAs primate shipments from Cambodia. The subpoena stems from an investigation started by the USDOJ last November regarding a smuggling ring illegally smuggling primates to the US from Cambodia. Because Cambodia is a major supplier of NHPs, this suspension will continue to exacerbate the problem of using NHPs to generate pre-clinical data. The ripple effect on drug discovery and clinical development could potentially pushback commencement of human clinical trials of promising new candidates for years – not to mention lost investment dollars for investors and delayed career progression for researchers.
"The macaque is already recognized as an endangered species by the International Union for the Conservation of Nature," U.S. Attorney for the Southern District of Florida Juan Antonio Gonzalez said in a statement. "The practice of illegally taking them from their habitat to end up in a lab is something we need to stop. Greed should never come before responsible conservation."
Organoids come to the rescue
Fortunately, all hope is not lost. Organoid technology, which has been developing rapidly, has become a cost effective alternative with several advantages over animal models including speed and cost. Late last year FDA removed the requirements for animal testing before moving into clinical testing which has made organoids a very attractive proposition.[1] Biotech players who can quickly position themselves to take advantage of this convergence by adopting organoids over traditional and lengthy animal model route can fast track their drug candidates into clinical trials. With the right platform in place, organoids can be ready for testing within a few weeks. This means that relevant safety and efficacy IND enabling data to commence clinical trials may be ready in a matter of months.
(Pic source: https://www.news-medical.net/news/20200515/First-detailed-study-of-the-epigenetic-landscape-of-human-tumor-organoids.aspx)
Fresh Wind Biotech is offering “one-stop shopping experience” for organoid research services
We were a group of young and bright PhD students at Baylor College of Medicine with the same inspiration of improving the outcomes of cancer patients. Now we grow up to a group of experienced and intelligent scientists, investors and entrepreneurs, aiming to realize our dreams of bringing effective and safe personalized therapies to cancer patients.
Indeed, Fresh Wind Biotech (FWB) has been at the forefront in using tumor organoids for its own clinical TIL therapy studies. Recently FWB has started granting access to its extensive organoid platform to the oncology research and biotech community through its organoid service. Using FWB’s platform, researchers have access to tumor organoids from a variety of cancer indications for use on their projects – a one stop and rapid solution that many researchers are finding appealing to the alternatives. Often times tumor organoids can be ready in 3-5 weeks.
For researchers, using an organoid service is an easy alternative to trying to grow organoids in-house. It allows them to short circuit many of the bottlenecks. These include procuring tumor samples which involve both locating tumor sources and obtaining IRB approval, as well as setting up the resources and experience to successfully grow organoids consistently. This trial and error divert time and focus away from their primary research purpose. For researchers focused on obtaining results sooner, outsourcing organoid production is an easy choice.
Savvy researchers and biotech players who are able to leverage these available services will be moving their projects to the next level at speed. More information about our tumor organoid service is available here.