TCR-engineered T (TCR-T) cell therapy is an innovative approach that harnesses the power of our immune system to fight against various diseases, including cancer. It involves genetically modifying T cells, a type of white blood cell, to express T cell receptors (TCRs) that target specific antigens found in cancer cells. One of the key advantages of TCR-T cell therapy is its ability to target a wide range of tumor antigens. Unlike the chimeric antigen receptor (CAR) T cell therapy that primally recognize and attack cellular surface antigens, TCR-T cells can target intracellular antigens as well, allowing for broader applicability. This flexibility makes it a promising treatment option for patients with solid tumors.
Clinical Trials of TCR-T
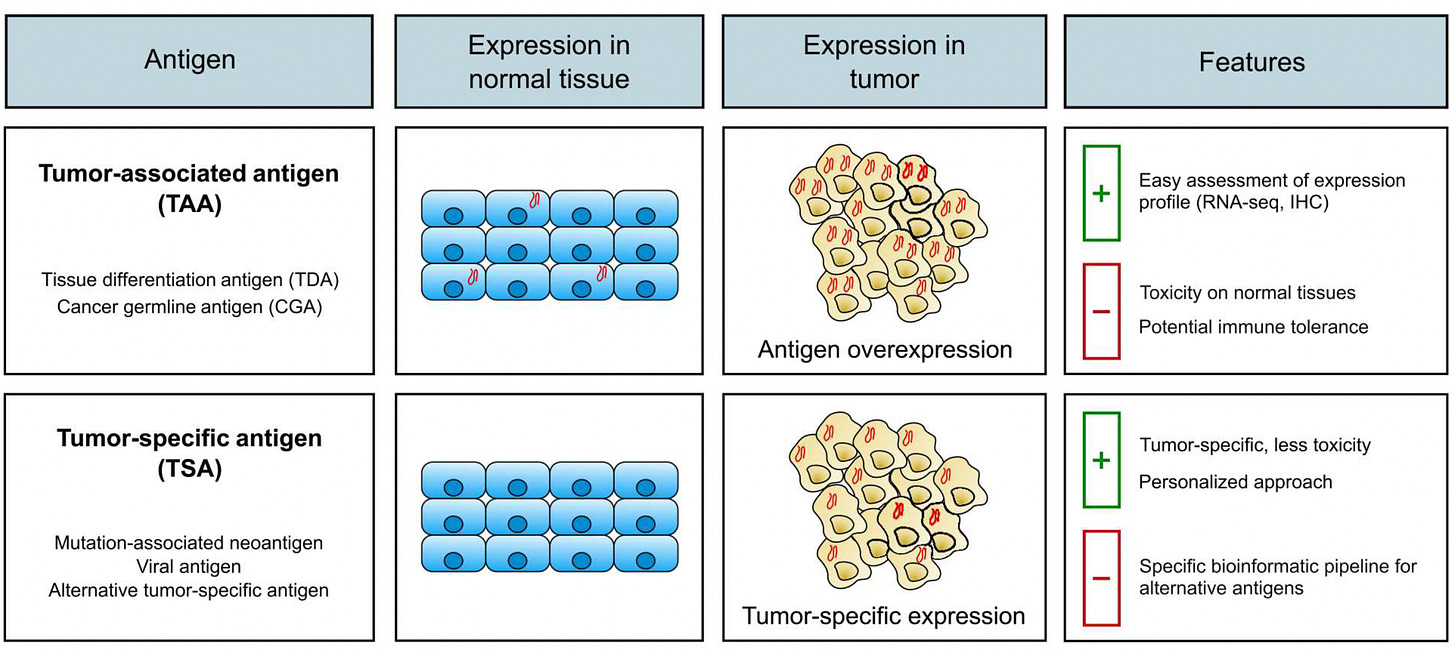
Clinical trials, summarized in the table, investigating TCR-T cell therapy for solid tumors have demonstrated promising outcomes. These trials have focused on two primary classes of antigens: tumor-associated antigens (TAAs) and tumor-specific antigens (TSAs).
TAAs, which are shared among patients, present attractive therapeutic targets. However, their expression in normal tissues, albeit at low levels, may lead to on-target off-tumor toxicity. On the other hand, TSAs, also known as neoantigens, result from tumor-specific mutations and are ideal targets for immunotherapies. A recent approach involves screening for TCRs that recognize shared neoepitopes derived from frequently mutated driver genes. For example, targeting a p53R175H neoepitope in a breast cancer patient using TCR-T cells resulted in a partial response with limited toxicity [1]. The pros and cons of TAAs and TSAs are illustrated as below, providing insights into their respective advantages and challenges in TCR-T cell therapy for solid tumors.
(The picture is from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9931212/figure/F1/)
TCR Identification
Identifying antigen-specific TCRs is a significant challenge in TCR-T cell therapy due to the intricate interaction between the TCR and the peptide-MHC complex. The conventional method involves antigen stimulation of T cells, followed by sorting and amplification of epitope-specific T cells in vitro. The TCR sequences are then determined through TCRα and TCRβ sequencing of isolated T cells after rapid PCR amplification.
The recent advancement in single-cell sequencing technology, specifically focused on TCR sequences (scTCR-seq), has greatly facilitated the identification of TCRs from primed T cells. This method allows for the identification of TCR sequences from T cells expressing high levels of activation markers after antigenic stimulation, thereby selecting highly functional TCRs in the process. Additionally, researchers, such as Dr. Rosenberg's lab, have identified gene expression signatures of antitumor neoantigen-reactive T cells from metastatic human tumors using single-cell transcriptome analysis [2]. Combining scTCR-seq with single-cell expression signatures enables faster and more accurate identification of antigen-reactive TCRs.
Fresh Wind Biotechnologies, Inc. developed a proprietary data analysis platform for precise and efficient TCR identification.
Other Challenges of TCR-T Cell Therapy
Besides identification and selection of suitable tumor antigens and reactive TCRs, other challenges remain, including:
· Toxicity assessment: Assessing and managing toxicity risks, including both on-target off-tumor and off-target off-tumor (cross-reactivity) effects.
· Optimizing T cell engineering: Exploring viral transduction, gene editing (e.g., CRISPR/Cas9), and improving manufacturing processes to enhance the efficiency and safety of T cell engineering.
· Tumor resistance mechanisms: Overcoming hurdles like target antigen downregulation or loss, as well as the immunosuppressive tumor microenvironment, which can limit the effectiveness of TCR-T cell therapies. Investigating combination therapies with immune checkpoint inhibitors to overcome tumor resistance.
Overall, addressing these challenges is crucial to improve the safety and efficacy of TCR-T cell therapy.
In Summary, TCR-T cell therapy has shown remarkable promise in treating solid tumors, even those that have limited responses to current immunotherapies. Although this therapeutic approach is complex, addressing challenges through improved selection of tumor-specific antigens and optimizing TCR identification and T cell engineering can enhance antitumor efficacy while reducing toxicity. Like next-generation CAR-T cells, TCR-T cell therapy has the potential to revolutionize the treatment landscape for cold tumors and address a significant unmet medical need.
References:
1. Malekzadeh P, Pasetto A, Robbins PF, Parkhurst MR, Paria BC, Jia L, Gartner JJ, Hill V, Yu Z, Restifo NP, Sachs A, Tran E, Lo W, Somerville RP, Rosenberg SA, Deniger DC. Neoantigen screening identifies broad TP53 mutant immunogenicity in patients with epithelial cancers. J Clin Invest. 2019 Mar 1;129(3):1109-1114. doi: 10.1172/JCI123791. Epub 2019 Feb 4. PMID: 30714987; PMCID: PMC6391139.
2. Lowery FJ, Krishna S, Yossef R, Parikh NB, Chatani PD, Zacharakis N, Parkhurst MR, Levin N, Sindiri S, Sachs A, Hitscherich KJ, Yu Z, Vale NR, Lu YC, Zheng Z, Jia L, Gartner JJ, Hill VK, Copeland AR, Nah SK, Masi RV, Gasmi B, Kivitz S, Paria BC, Florentin M, Kim SP, Hanada KI, Li YF, Ngo LT, Ray S, Shindorf ML, Levi ST, Shepherd R, Toy C, Parikh AY, Prickett TD, Kelly MC, Beyer R, Goff SL, Yang JC, Robbins PF, Rosenberg SA. Molecular signatures of antitumor neoantigen-reactive T cells from metastatic human cancers. Science. 2022 Feb 25;375(6583):877-884. doi: 10.1126/science.abl5447. Epub 2022 Feb 3. PMID: 35113651; PMCID: PMC8996692.